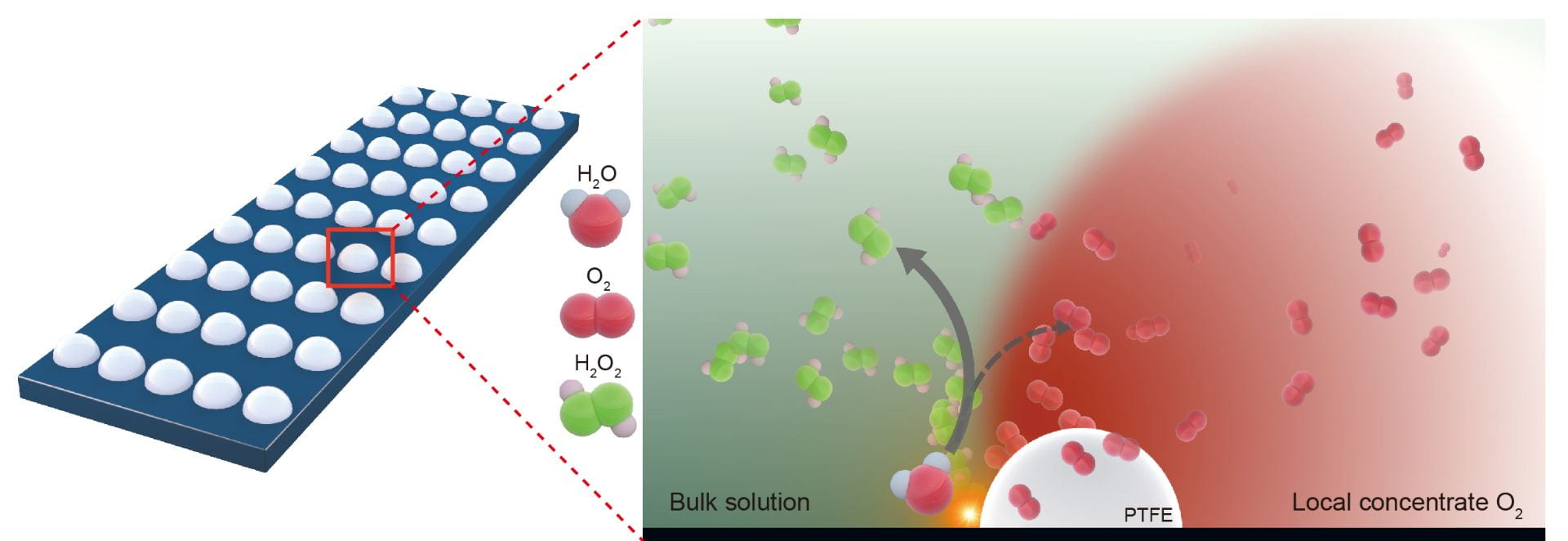
Congratulations to Chuan and our close collaboratiors, Seoin, Stefan, Samira, and Karen on the publication of our water oxidation to hydrogen peroxide work in Nature Catalysis! This is a very interesting finding out of an unexpected experimental result. Typically, to deliver a better performance in electrochemical water oxidation, the electrode needs to be as hydrophilic as possible to allow more contacts between catalysts and water. People rarely use hydrophobic electrodes in water oxidation or reduction reactions, but only in gas reduction or oxidation reactions involving triple phase boundaries. In this study, we surprisingly discovered that , by using a very hydrophobic electrode in water oxidation, its two electron reaction pathway to H2O2 was greatly enhanced. Pay attention to every details in experiments and they will open a new world to us!
Abstract: Electrochemical two-electron water oxidation is a promising route for renewable and on-site H2O2 generation as an alternative to the anthraquinone process. However, it is currently restricted by low selectivity due to strong competition from the traditional four-electron oxygen evolution reaction, as well as large overpotential and low production rates. Here we report an interfacial engineering approach, where by coating the catalyst with hydrophobic polymers we confine in situ produced O2 gas to tune the water oxidation reaction pathway. Using carbon catalysts as a model system, we show a significant increase of the intrinsic H2O-to-H2O2 selectivity and activity compared to that of the pristine catalyst. The maximal H2O2 Faradaic efficiency was enhanced by sixfold to 66% with an overpotential of 640 mV, under which a H2O2 production rate of 23.4 µmol min−1 cm−2 (75.2 mA cm−2 partial current) was achieved. This approach was successfully extended to nickel metal, demonstrating the wide applicability of our local gas confinement concept.

Indeed its a great contribution in the field of electrocatalysis for water splitting and CO2 reduction. Congratulation to entire team and specially Dr. Haitong wang.
I am attracted by this fulling of passion and motivation group. Even though our parents and elder relatives told us we should be “down to earth”, how to say, sometimes, what truly promoting the progress of the entire humanity is “Looking at the stars”!